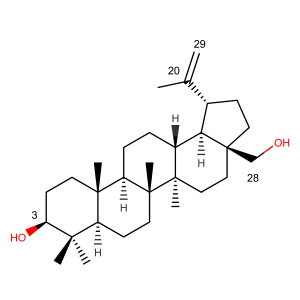
Betulin Numbering and Nomenclature
The numbering of betulin and betulinic acid is based on well-defined guidelines developed by IUPAC. The set of rules defines to first identify the base structure. Both, betulin and betulinic acid share the same molecular scaffold that belongs to the group of pentacyclic triterpenes. This group is based on lupane. By IUPAC definition numbering of betulin is the same as lupane.
Naming structures using this semi-trivial nomenclature e.g. lupane, that already defines overall structure and stereochemistry of the whole scaffold. Carbon numbering that is shown in the scheme can be used to identify the position of substituents or functional groups, that are attached by different chemical modifications. Stereo-chemistry of these modifications can be shown by using Greek letters.
- α is used for substituent being anti- to methyl group at position C(17).
- β is used for substituents being cis- to methyl group at position C(17).
For example, the IUPAC name of betulin would be 3β-Hydroxylup-20(29)-ene:
- 3β showing the location of the secondary hydroxyl- group;
- 20(29)-en showing the location of an unsaturated double bond in betulin;
- 28-ol showing the location of the primary hydroxyl- group.
IUPAC name of betulinic acid would be 3β-Hydroxylup-20(29)-en-28-oic acid and so on.
The mol file of betulin structure file and useful for any derivative drawing that is suitable for all structure drawing programs can be downloaded here.